Pharmaceutical Contract Manufacturing Service
of Nipro Group
Pharmaceutical Contract Manufacturing Top class in Japan
Winning trust with the company's creative and flexible abilities
Nipro Group has widened its operations as an all-around manufacturer of contract pharmaceutical products, based on its creative development power and production technologies, and has become top class manufacturer of contract pharmaceutical products in Japan. Repeated orders from numerous pharmaceutical companies are the best evidence of the trust placed in Nipro Pharma.
One-stop shop for all your needs
All-round contract manufacturing
Comprehensive Contract Manufacturing Services
- Contract manufacturing services for all dosage forms, including oral products,injectables and external preparations.
- Nipro Group,in addition to its well-known production capacity and technological competence,has an unrivaled capacity for development.
- Quick, flexible development ranging from pharmaceutical formulations to containers according to the requirements of customers.
High-Value-Added Pharmaceutical Formulations and Generic Drugs
- Proposal and development of high-value-added pharmaceutical formulations, including kit formulations, orally disintegrating tablets, DDS-applied products, TTS-applied products based on Nipro's highly advanced formulation technologies.
- Various supports for formulation development, marketing approval application, etc. based on Nipro's formulation tecnologies and project-management expertise.
Risk Management
- Commitment to BCP(Business Continuty Planning) -
- Highly reliable system established for pharmaceutical supply with the dispersion of production bases(the Nipro Group network)
- Securement of secondary sources for active pharmaceutical ingredients.
Global CDMO
- Contract manufacturing of pharmaceutical products for overseas markets in compliance with GMP(c-GMP, EU-GMP,JGMP)and PIC/S.
- Comprehensive support system for overseas customers seeking entry to the Japanese market.
- Nipro Pharma Vietnam offers products with high quality level at internationally competitive prices.
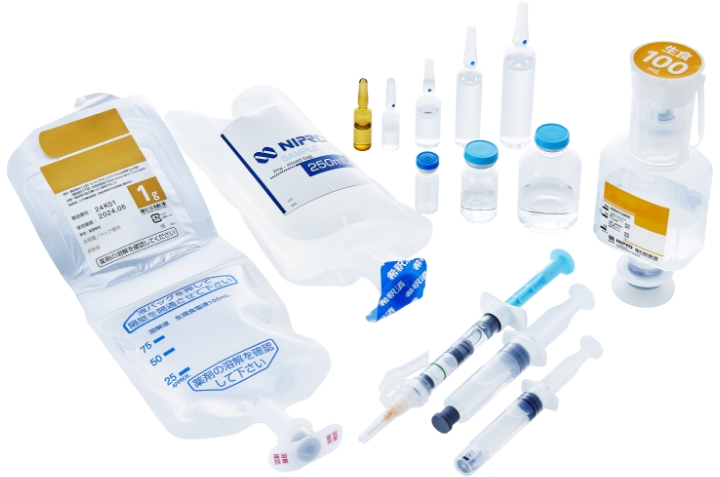
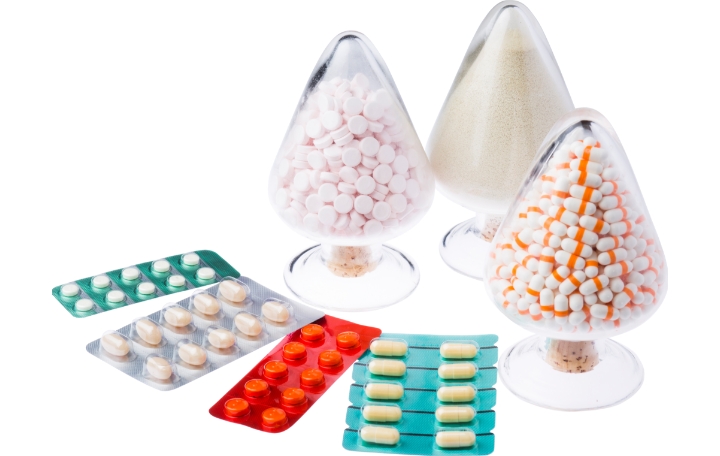
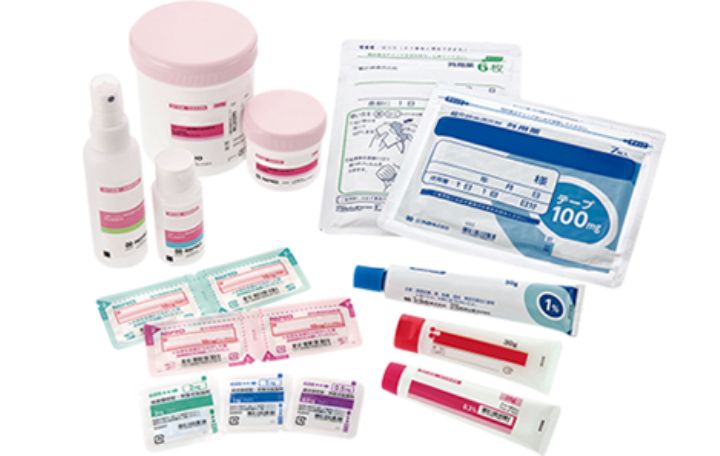
Dosage Forms that Nipro Pharma can provide
Nipro Pharma has production abilities and facilities for all categories including injectables, oral products, and external preparations. We will further enhance our production abilities to respond to the new and diverse needs of pharmaceutical companies.
Injectables
- Ampoules(Glass・Plastics)
- Vials(Liquid・Powder・Lyophilized products)
- Pre-filled syringes(Glass・Plastics)
- Dual chamber pre-filled syringes(Glass)
- Dual bags(Liquid / Powder)
- Dual bags(Liquid / Liquid)
- Half-type kits
- Pre-mixed bags