
Odate Plant
Main Plant for kits for injectables; equipped with separate dedicated buildings for respective drugs as well as a cutting-edge production line for external preparations
Producing pre-filled syringes, double bags (PLW®), and other kits for injectables in general, is one of the greatest strengths of Nipro Pharma. Thorough containment measures have been taken for highly reactive agents including anticancer drugs, antibiotics, and steroids that are showing high market growth. Measures such as constructing separate buildings for each type of drug are taken. For external preparations, we introduced a cutting-edge production line for solvent type tapes, thoroughly ensuring a quality assurance system capable of meeting the GMPs requirements in Europe, Japan and the United States.
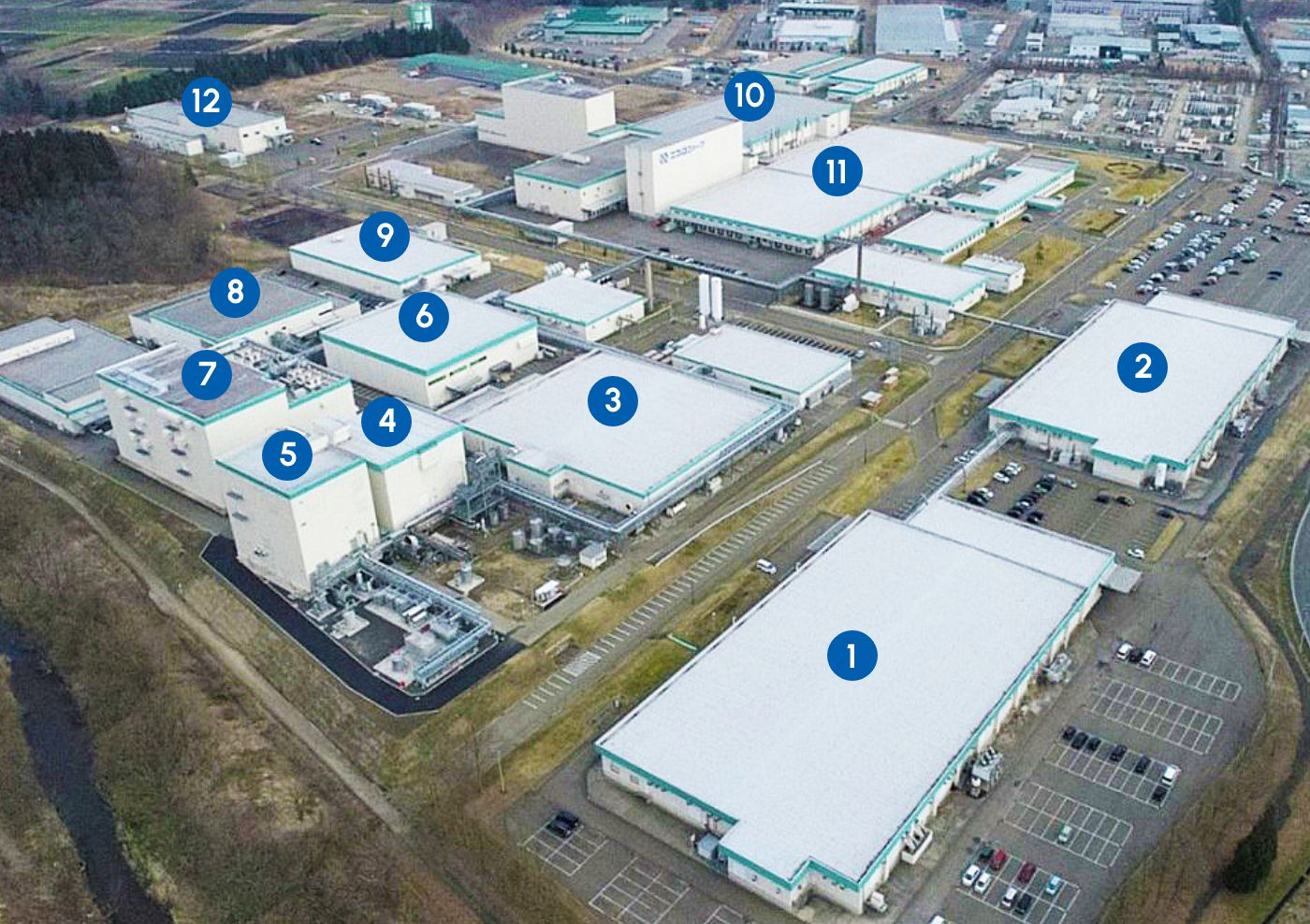
- Cephem DB production bldg.
- Penicillin DB production bldg.
- Cephem (freeze-dried/powder) production bldg.
- Cephem drug substance production bldg.
- Cephem API Synthesis bldg.
- Carbapenem production bldg.
- Hormone production bldg.
- Anticancer drug production bldg.
- Steroid production building
- Pre-filled syringe production bldg.
- Biomedicine production building
Dialysis fluid replacement DB production bldg.(Liquid/liquid line) - Tapes producing bldg.
Odate Plant Video Introduction
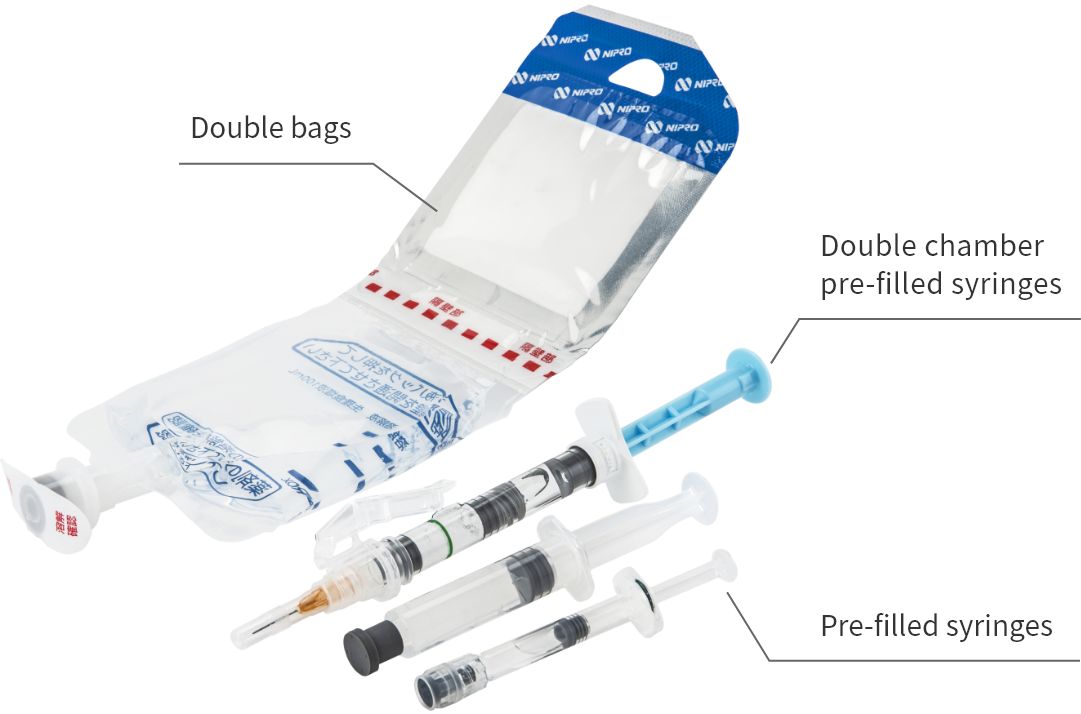
Produces injectable drug kits in general
At this plant, we produce pre-filled syringes (PFS®), i.e., syringes pre-filled with drug solution, double chamber bags (PLW®)providing drug solutions combined with dissolving solutions, as well as kit preparations including double chamber pre-filled syringes, involving the application of double chamber bags to syringes, thereby contributing to improved sterility assurance, the prevention of medical mistakes and efficiency in medical settings.

High potent drugs are produced in dedicated bldg.
To reduce risks to handle high potent material and to assure safety of the workers, we use high containment technologies and make the buildings completely isolated.
We produce bio pharmaceuticals and anticancer drugs in response to the increasing production requests from customer companies. In the anticancer drug production bldg., where intensified safety measures are required, we use robots inside the isolator to achieve full automation. We also make suggestions concerning countermeasures against bottle breakage, so that damage to glass containers will be minimized.

Diverse and enhanced high-functional production lines
Here, we have a variety of different equipment to produce high-quality pharmaceutical products. On the pre-filled syringe production line, we have established an aseptic operation combined with quality assurance, based on integrated production through to the final packaging. The production lines for API are also equipped with refining facilities to produce high-quality pharmaceutical products.
Quick facts about the Odate Plant
| Location |
5-7, Niida Aza Maedano, Odate-Shi, Akita, 018-5751, Japan |
|---|---|
| TEL |
+81-186-44-8650 |
| FAX |
+81-186-44-8690 |


